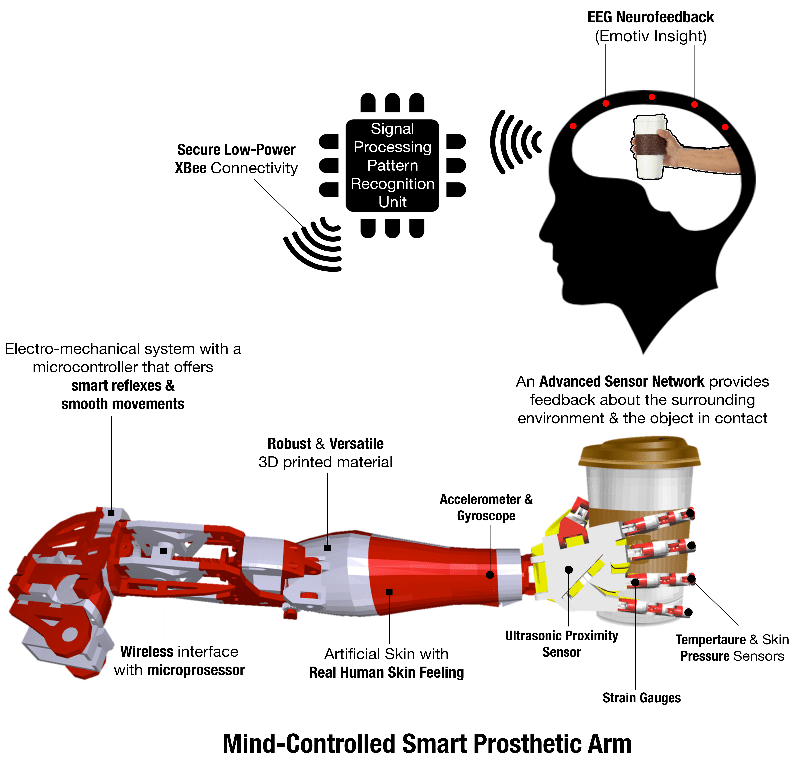
Thought-Controlled Wireless Electronic Prosthesis
Stanford researchers have been working for years to advance a technology that could one day help people with paralysis regain use of their limbs and enable amputees to use their thoughts to control prostheses and interact with computers.

The team has been focusing on improving a brain-computer interface, a device implanted beneath the skull on the surface of a patient's brain. This implant connects the human nervous system to an electronic device that might, for instance, help restore some motor control to a person with a spinal cord injury, or someone with a neurological condition like amyotrophic lateral sclerosis, also called Lou Gehrig's disease.
The current generation of these devices record enormous amounts of neural activity, then transmit these brain signals through wires to a computer. But when researchers have tried to create wireless brain-computer interfaces to do this, it took so much power to transmit the data that the devices would generate too much heat to be safe for the patient.
Now, a team led by electrical engineers and neuroscientists Krishna Shenoy, PhD, and Boris Murmann, PhD, and neurosurgeon and neuroscientist Jaimie Henderson, MD, have shown how it would be possible to create a wireless device, capable of gathering and transmitting accurate neural signals, but using a tenth of the power required by current wire-enabled systems. These wireless devices would look more natural than the wired models and give patients a freer range of motion.
Graduate student Nir Even-Chen and postdoctoral fellow Dante Muratore, PhD, describe the team's approach in a Nature Biomedical Engineering paper.
In essence, the team has circumvented today’s painstaking process of tracking the activity of individual neurons in favour of decoding neural activity in the aggregate. Each time a neuron fires it sends an electrical signal — known as a “spike” — to the next neuron down the line. It’s the sort of intercellular communication that turns a notion in the mind into muscle contraction elsewhere in the body. “Each neuron has its own electrical fingerprint and no two are identical,” says Eric Trautmann, a postdoctoral researcher in Shenoy’s lab and first author of the paper. “We spend a lot of time isolating and studying the activity of individual neurons.”
Neuroscientists call that process “spike sorting,” and it must be done for every neuron in every experiment, an endeavour that eats up many thousands of hours of researcher time each year and is only going to get more time consuming as scientists build implants with greater numbers of electrodes. Indeed, researchers estimate that sensors will have 1,000 or more electrodes — up from 100 today — at which point, it would take a neuroscientist 100 hours or more to sort the spikes by hand for every experiment.
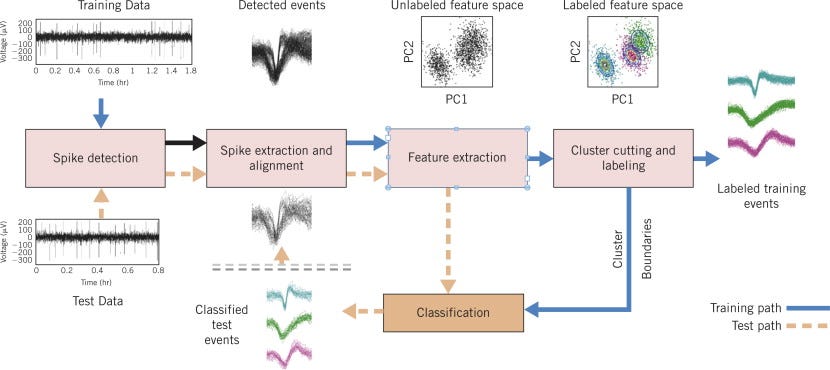
To record the activity of a number of neurons without the complexity of spike sorting, the researchers borrowed a theory from statistics that suggested how they could uncover patterns of brain activity even when several neurons are recorded on a single electrode. They then demonstrated their approach experimentally. They used a new type of electrode that was designed to pick up brain signals in mice and adapted this technology to record the brain signals of rhesus monkeys. They recorded hundreds of neurons at the same time and showed that they could get an accurate portrait of the monkey’s brain activity without spike sorting.
The team's electrical engineers then designed the circuitry that would enable a future, wireless brain-computer interface to process and transmit these carefully identified and isolated signals, using less power and thus making it safe to implant the device on the surface of the brain.
To test their idea, the researchers collected neuronal data from three nonhuman primates and one human participant in a (BrainGate) clinical trial.
As the subjects performed movement tasks, such as positioning a cursor on a computer screen, the researchers took measurements. The findings validated their hypothesis that a wireless interface could accurately control an individual's motion by recording a subset of action-specific brain signals, rather than acting like the wired device and collecting brain signals in bulk.
The next step will be to build an implant based on this new approach and proceed through a series of tests toward the ultimate goal.
An Article By: Ignite Team
DISCLAIMER
Shaastra TechShots’ publications contain information, opinions and data that Shaastra TechShots considers to be accurate based on the date of their creation and verified sources available at that time. It does not constitute either a personalized opinion or a general opinion of Shaastra or IIT Madras. The information provided comes from the best sources, however, Shaastra TechShots cannot be held responsible for any errors or omissions that may emerge.



